Understanding Transition Metal Complexes:Back-bonding with CO and NO
1. Rygbinding: Dette er den vigtigste drivkraft. Transition metals in these low oxidation states have a high density of electrons in their d-orbitals. Ligands like CO and NO possess empty antibonding π* orbitals.
* The filled d-orbitals of the metal can donate electron density into the empty π* orbitals of the ligand, forming a π-backbond .
* This back-bonding interaction strengthens the metal-ligand bond significantly.
2. Synergisk binding: This refers to the combined effect of σ-donation and π-backbonding.
* The ligand (CO or NO) donates electron density to the metal through a σ-bond.
* This donation makes the metal more electron-rich, facilitating the back-donation process.
3. Stabilitet: π-backbonding-interaktionen fører til:
* Øget elektrontæthed: The metal center gains electron density, leading to increased stability.
* Svækkede ligandbindinger: The back-donation into the π* orbitals weakens the C-O and N-O bonds in CO and NO, respectively, increasing their reactivity.
4. Elektronisk konfiguration: Transition metals in low oxidation states often have a d 8 eller d 10 electronic configuration, which favors complex formation with strong π-acceptor ligands like CO and NO.
5. Ligandegenskaber: CO og NO er begge stærke π-acceptorligander. Their ability to accept electron density from the metal is crucial for the back-bonding interaction.
Eksempel:
* I nikkelcarbonyl (Ni(CO)4 ), er nikkelatomet i en nuloxidationstilstand.
* CO-liganderne donerer elektroner til nikkel gennem σ-bindinger og modtager tilbagedonation fra nikkelens fyldte d-orbitaler til deres π*-antibindings-orbitaler.
* This strong back-bonding makes nickel carbonyl a very stable compound.
Konklusion:
Kombinationen af tilbagebinding, synergisk binding og de gunstige elektroniske konfigurationer af overgangsmetaller i lave oxidationstilstande gør kompleksdannelse med ligander som CO og NO yderst favoriseret. These complexes are often very stable due to the strong metal-ligand bonds formed through back-bonding.
 Varme artikler
Varme artikler
-
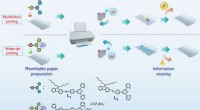 Specialpapir kan slettes og udskrives på flere gangeSkematisk illustration af flerfarve- og vandtryk. Kemiske strukturer af L 1 , L2 og L3, og udskrivnings- og sletteprocesserne for det konstruerede genskrivbare papir. Kredit: Naturkommunikation (2017
Specialpapir kan slettes og udskrives på flere gangeSkematisk illustration af flerfarve- og vandtryk. Kemiske strukturer af L 1 , L2 og L3, og udskrivnings- og sletteprocesserne for det konstruerede genskrivbare papir. Kredit: Naturkommunikation (2017 -
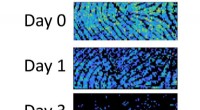 Rester i fingeraftryk indeholder spor af deres alderNiveauer af umættet triacylglycerol falder i fingeraftryk fra et individ fra dag 0 (øverst) til dag 1 (midten) og dag 3 (nederst). Kredit:Tilpasset fra Analytisk kemi 2020, DOI:10.1021/acs.analchem.
Rester i fingeraftryk indeholder spor af deres alderNiveauer af umættet triacylglycerol falder i fingeraftryk fra et individ fra dag 0 (øverst) til dag 1 (midten) og dag 3 (nederst). Kredit:Tilpasset fra Analytisk kemi 2020, DOI:10.1021/acs.analchem. -
 Kemiker viser, at intermolekylære interaktioner kan opnå hidtil ukendte dimensionerKredit:The Journal of Physical Chemistry Letters Intermolekylære interaktioner er de kræfter, der vedrører molekyler. Generelt strækker disse interaktioner sig næppe ud over molekylernes grænser.
Kemiker viser, at intermolekylære interaktioner kan opnå hidtil ukendte dimensionerKredit:The Journal of Physical Chemistry Letters Intermolekylære interaktioner er de kræfter, der vedrører molekyler. Generelt strækker disse interaktioner sig næppe ud over molekylernes grænser. -
 Forskere opdager nye ikke-klæbende gelerBillede af ikke-klæbende gel, der tager ved hjælp af avancerede 3D-lysmikroskopiteknikker. Kredit:University of Bristol Forskere fra University of Bristol og Université Paris-Saclay har opdaget en
Forskere opdager nye ikke-klæbende gelerBillede af ikke-klæbende gel, der tager ved hjælp af avancerede 3D-lysmikroskopiteknikker. Kredit:University of Bristol Forskere fra University of Bristol og Université Paris-Saclay har opdaget en
- Hvilke karakteristika kunne bruges til at klassificere dyr i forskellige grupper?
- Palæstinensiske landmænd nyder godt af Birmingham vandteknologiprojekt
- Ekspertguide til at holde dig bidefri denne sommer
- Sådan beregnes MTBF
- Under isen
- Undersøgelse finder, at saltnanopartikler er giftige for kræftceller