Calculating Hydronium Ion Concentration from pH:A Step-by-Step Guide
Forstå forholdet
* pH: Et mål for surhedsgraden eller alkaliniteten af en opløsning. It's defined as the negative logarithm (base 10) of the hydrogen ion (H+) concentration.
* H3O+: The hydronium ion, which is essentially a hydrogen ion (H+) bonded to a water molecule (H2O). In aqueous solutions, H+ ions exist primarily as H3O+ ions.
Ligningen
The relationship between pH and H3O+ concentration is:
pH =-log[H3O+]
Løser for [H3O+]
1. Omarranger ligningen:
[H3O+] =10^(-pH)
2. Tilslut pH-værdien:
[H3O+] =10^(-3,120)
3. Beregn:
[H3O+] ≈ 7,58 x 10^-4 M
Svar: The molar concentration of H3O+ in the cola is approximately 7.58 x 10^-4 M.
Sidste artikelSvovlatomtal i H2S + SO2 → H2-reaktion:en trin-for-trin guide
Næste artikelBeregning af pH af en bufferopløsning (HNO₂ &NaNO₂)
 Varme artikler
Varme artikler
-
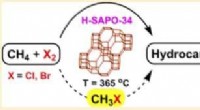 At reducere fodaftrykket af en drivhusgas, der er mere potent end kuldioxidUSC-forskere brugte H-SAPO-34 til direkte at omdanne metan til ethylen og propylen. Kredit:G. K. Surya Prakash USC-forskere har låst op for en ny, mere effektiv vej til at omdanne metan - en poten
At reducere fodaftrykket af en drivhusgas, der er mere potent end kuldioxidUSC-forskere brugte H-SAPO-34 til direkte at omdanne metan til ethylen og propylen. Kredit:G. K. Surya Prakash USC-forskere har låst op for en ny, mere effektiv vej til at omdanne metan - en poten -
 Biofysikere målretter mod mekanisme, der gør Zika -virus så farligtKredit: Naturens kemiske biologi (2021). DOI:10.1038/s41589-021-00829-z En ny undersøgelse foretaget af biofysikere fra University of Alberta har afsløret, hvordan en sjælden struktur dannes i RN
Biofysikere målretter mod mekanisme, der gør Zika -virus så farligtKredit: Naturens kemiske biologi (2021). DOI:10.1038/s41589-021-00829-z En ny undersøgelse foretaget af biofysikere fra University of Alberta har afsløret, hvordan en sjælden struktur dannes i RN -
 Forskere har opdaget nye forbindelser med herbicid potentiale fra havsvampOlesya Zhuravleva, Ph.D., Leder af Laboratoriet for biologisk aktive forbindelser, FEFU Naturvidenskabelige Skole. Kredit:FEFUs pressekontor Forskere ved Far Eastern Federal University (FEFU) og G
Forskere har opdaget nye forbindelser med herbicid potentiale fra havsvampOlesya Zhuravleva, Ph.D., Leder af Laboratoriet for biologisk aktive forbindelser, FEFU Naturvidenskabelige Skole. Kredit:FEFUs pressekontor Forskere ved Far Eastern Federal University (FEFU) og G -
 Fremstillingen af en krystalflipperKredit:CC0 Public Domain Forskere fra Hokkaido-universitetet har fremstillet en krystal, der automatisk vipper frem og tilbage, mens de ændrer sine vendemønstre som reaktion på lysforholdene. Dere
Fremstillingen af en krystalflipperKredit:CC0 Public Domain Forskere fra Hokkaido-universitetet har fremstillet en krystal, der automatisk vipper frem og tilbage, mens de ændrer sine vendemønstre som reaktion på lysforholdene. Dere
- Hvilken energitransformation opstår, når en baseløber glider sikkert til tredje base?
- Lille, præcist og overkommeligt gyroskop til navigation uden GPS
- Hackere retter sig mod Airbus -leverandører i jagten på kommercielle hemmeligheder
- Hvad er en enhed, der konverterer elektrisk energi til lineær kraft?
- Hvad er energikonverteringen i en tørringsmaskine?
- Hvad er den kemiske formel for jernjod?