Understanding CO₂ Density:From Gases to Dry Ice
Af Jack Brubaker – Opdateret 30. august 2022
Vladimir_Timofeev/iStock/GettyImages
Kuldioxid (CO₂) cirkulerer i Jordens atmosfære med omkring 0,033 procent. It forms during animal respiration and hydrocarbon combustion. Unlike most gases, CO₂ does not liquidate at ambient temperatures; it sublimates—transiting directly from solid to gas.
Density Fundamentals
Density is the mass of a substance per unit volume, expressed in grams per milliliter (g/mL) or grams per cubic centimeter (g/cc). For gases, the metric is usually g/mL due to the small values involved.
Gasformig CO₂
At standard conditions (0 °C, 1 atm), CO₂ has a density of 0.001977 g/mL. This is marginally higher than air’s 0.001239 g/mL, making CO₂ slightly heavier and more prone to accumulate in low‑lying areas.
Fast CO₂ (tøris)
In its solid form—commonly called dry ice—CO₂’s density rises to 1.56 g/mL under standard conditions. For context, liquid water’s density is about 1.00 g/mL, so dry ice sinks when introduced to water.
These values are corroborated by data from the NIST Chemistry WebBook and other reputable chemical databases.
Understanding CO₂ density is essential for applications ranging from climate science to industrial processes.
 Varme artikler
Varme artikler
-
 Opsugning af lægemidler og produkter til personlig pleje fra vandKredit:American Chemical Society Medicin, der udskilles i urinen eller dumpes i toilettet, kan ende i vandforsyningen, ligesom lotioner eller kosmetik, der vasker kroppen af og går ned i vasken
Opsugning af lægemidler og produkter til personlig pleje fra vandKredit:American Chemical Society Medicin, der udskilles i urinen eller dumpes i toilettet, kan ende i vandforsyningen, ligesom lotioner eller kosmetik, der vasker kroppen af og går ned i vasken -
 Forskere skaber ny form for dyrket kødKredit:Pixabay/CC0 Public Domain McMaster-forskere har udviklet en ny form for dyrket kød ved hjælp af en metode, der lover mere naturlig smag og tekstur end andre alternativer til traditionelt kø
Forskere skaber ny form for dyrket kødKredit:Pixabay/CC0 Public Domain McMaster-forskere har udviklet en ny form for dyrket kød ved hjælp af en metode, der lover mere naturlig smag og tekstur end andre alternativer til traditionelt kø -
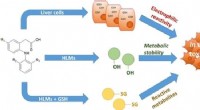 Subtile ændringer i kemisk struktur kan påvirke lægemiddeltoksicitetDerivater af lumiracoxib blev syntetiseret for at forstå forskellige kemiske gruppers bidrag til toksicitet. Forbindelserne blev testet i humane leverceller, og også med og uden tilstedeværelse af glu
Subtile ændringer i kemisk struktur kan påvirke lægemiddeltoksicitetDerivater af lumiracoxib blev syntetiseret for at forstå forskellige kemiske gruppers bidrag til toksicitet. Forbindelserne blev testet i humane leverceller, og også med og uden tilstedeværelse af glu -
 Kemiker studerer elektriske felter, mikrofluidik til forbedring af dialyteteknologiJoseph Banovetz, Beatrise Berzina og Robbyn Anand, venstre til højre, oprette en prototype -enhed til at teste separationsydelse i blodplasma. De bruger et fluorescensmikroskop til at følge to sporsto
Kemiker studerer elektriske felter, mikrofluidik til forbedring af dialyteteknologiJoseph Banovetz, Beatrise Berzina og Robbyn Anand, venstre til højre, oprette en prototype -enhed til at teste separationsydelse i blodplasma. De bruger et fluorescensmikroskop til at følge to sporsto
- Hvordan bruger vi energien?
- Hvordan bliver plutoner synlige på jordoverfladen, hvis de dannes under jorden?
- Nukleolus og endoplasmatisk retikulum:Delte cellulære komponenter
- Beskriv de tre typer små partikler i et atom og forklar, hvordan de er arrangeret?
- Hvorfor vokser mange planter i en regnskov?
- Hvad er en skovbrand?