SO2 Molecular Geometry:Understanding the Bent Shape
* Centralatom: Svovl (S) er det centrale atom.
* Valenselektroner: Sulfur has 6 valence electrons, and each oxygen atom has 6 valence electrons.
* Binding: Sulfur forms double bonds with each oxygen atom, sharing two electrons with each oxygen.
* Ensomme par: Svovl har også et ensomt elektronpar.
* Elektronparrepulsion: The electron pairs (both bonding and lone pairs) around the sulfur atom repel each other. This repulsion leads to a bent shape, minimizing the repulsion between the electrons.
Therefore, the SO₂ molecule has a bent shape with an approximate bond angle of 119 degrees.
Sidste artikelIltpolaritet:Forståelse af skiftet fra O₂ til H₂O
Næste artikelForståelse af pH i destilleret vand:En neutral forklaring
 Varme artikler
Varme artikler
-
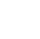
 Forskere afslører trinvise hydreringsmotiver af svovldioxidGrafisk abstrakt. Kredit:The Journal of Physical Chemistry Letters (2022). DOI:10.1021/acs.jpclett.2c01472 Et forskerhold ledet af prof. Jiang Ling og Zhang Zhaojun fra Dalian Institute of Chemical
Forskere afslører trinvise hydreringsmotiver af svovldioxidGrafisk abstrakt. Kredit:The Journal of Physical Chemistry Letters (2022). DOI:10.1021/acs.jpclett.2c01472 Et forskerhold ledet af prof. Jiang Ling og Zhang Zhaojun fra Dalian Institute of Chemical -
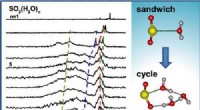
 Kemikere fremmer evnen til at kontrollere kemiske reaktionerValget af påvirkningsparameteren er grundlæggende for resultatet af kemisk reaktion, som visualiseret her baseret på eksperimenter. Ved nul indvirkningsparameter, reaktionen øverst viste reproducerbar
Kemikere fremmer evnen til at kontrollere kemiske reaktionerValget af påvirkningsparameteren er grundlæggende for resultatet af kemisk reaktion, som visualiseret her baseret på eksperimenter. Ved nul indvirkningsparameter, reaktionen øverst viste reproducerbar -
 Studer polyfarmakologi tidligere i lægemiddelopdagelsen, siger forskereKredit:CC0 Public Domain Polyfarmakologi - et lægemiddels evne til at påvirke mere end ét protein - bør studeres tidligt i lægemiddelopdagelsesforløbet, og konstant overvåget, da den kemiske struk
Studer polyfarmakologi tidligere i lægemiddelopdagelsen, siger forskereKredit:CC0 Public Domain Polyfarmakologi - et lægemiddels evne til at påvirke mere end ét protein - bør studeres tidligt i lægemiddelopdagelsesforløbet, og konstant overvåget, da den kemiske struk -
 Halvsyntetiske bakterier udskiller unaturlige proteinerForskere identificerede dette unaturlige basepar som værende optimalt til informationslagring i en semisyntetisk organisme. Kredit:Tilpasset fra Journal of the American Chemical Society 2019 , DOI
Halvsyntetiske bakterier udskiller unaturlige proteinerForskere identificerede dette unaturlige basepar som værende optimalt til informationslagring i en semisyntetisk organisme. Kredit:Tilpasset fra Journal of the American Chemical Society 2019 , DOI
- Foreslået metode til at få et atom til at udsende det samme lys som et andet atom
- Hvad kan Facebook have at skjule?
- Hvis celler multiplicerer, som de gør, når de dyrkes i vævskulturer, finder mitose sted T eller F…
- Vild fødesøgning tegner sig for næsten 250.000 kubikmeter produkter, der forbruges årligt i Zamb…
- Sådan beregnes T-testværdien
- En højhælet dinosaur, der gik på tæerne